The Green House Effect Lab
Pre-lab
What is the green house effect?
Green house effect- green house gases cause some radiation from the sun to be trapped as heat in the troposphere
Green house effect- green house gases cause some radiation from the sun to be trapped as heat in the troposphere
Parts of the Experiment
Independent Variable: what was contained in the beaker (ie. air, water, or soil?)
Dependent Variable: the increase of temperature within the allotted time
Controlled Variables: the time spent heating, the method of heating, and the concealment of the heat
Control Group: the beaker with only air contained inside
Experiment Groups: the beaker with water inside and the beaker with soil inside
Dependent Variable: the increase of temperature within the allotted time
Controlled Variables: the time spent heating, the method of heating, and the concealment of the heat
Control Group: the beaker with only air contained inside
Experiment Groups: the beaker with water inside and the beaker with soil inside
Hypothesis
If a beaker with air, a beaker with water, and a beaker with soil were all tested to determine which conditions harbor the quickest warming, then the experimental group with the soil will warm the quickest.
Problem
1. How quickly does the warming of the atmosphere occur?
2. What conditions cause the warming of the atmosphere?
2. What conditions cause the warming of the atmosphere?
Materials
-3 plastic bags
-3 500mL beakers
-3 thermometers
-1 heat lamp
-a stopwatch timer
-water
-soil
-3 500mL beakers
-3 thermometers
-1 heat lamp
-a stopwatch timer
-water
-soil
Procedure
1. Fill the beaker to the halfway point with the material being tested (leave empty for air).
2. Make sure the thermometers are set to room temperature for accuracy.
3. Set the thermometer with the bulb towards the material (towards the bottom of the beaker for air).
4. Cover the beaker with a plastic bag.
5. Simultaneously turn on the lamp and start the stopwatch timer.
6. Let the lamp heat the beaker for the set duration of time (1 minute the first loop and 2 minutes the second loop).
7. Turn off the lamp and stop the stopwatch timer.
8. Remove the plastic bag and thermometer and record the results.
9. Cycle through the beakers, allowing time for the materials to cool down to room temperature.
10. Create a results table and analyze.
2. Make sure the thermometers are set to room temperature for accuracy.
3. Set the thermometer with the bulb towards the material (towards the bottom of the beaker for air).
4. Cover the beaker with a plastic bag.
5. Simultaneously turn on the lamp and start the stopwatch timer.
6. Let the lamp heat the beaker for the set duration of time (1 minute the first loop and 2 minutes the second loop).
7. Turn off the lamp and stop the stopwatch timer.
8. Remove the plastic bag and thermometer and record the results.
9. Cycle through the beakers, allowing time for the materials to cool down to room temperature.
10. Create a results table and analyze.
Observations

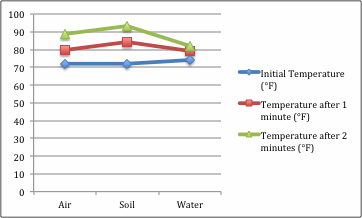
Throughout the experiment, it became apparent that the beaker with soil warmed up the fastest, followed by the beaker with air. For some reason, the thermometer that was used with the water read 74 degrees Fahrenheit at room temperature; this may have skewed the results. The beaker with water was not filled up to the halfway point, but somewhere around the 1/4-1/3 full. Because of the lack of materials, the thermometer used with the beaker filled with only air was farther away from the lamp. This experiment seems to have created a more accurate model of the warming of the atmosphere than other experiments that were tested.
Data Table and Graph
Analysis and Conclusions
The results of the experiment showed that the atmosphere with soil below it heated up the quickest. This reflected the hypothesis precisely. The experiment has shown that certain conditions do, in fact, effect the warming of the atmosphere. The predominant conditions that affected the experiment were the presence or absence of water or soil. This can correlate to the Earth's atmosphere by the presence or absence of lakes, rivers or oceans, and soil, clay, or dust. The reason the beaker with the soil heated up quicker may have been because some of the light was reflected off of the soil. The water and glass both let light pass through, instead of reflecting it off of its surface. The light reflected off of the soil probably heated the gases around it quicker. Another factor that may have affected the experiment is the distance of the thermometers from the light. In the soil beaker, the thermometer was the closest to the light because the soil is solid and the thermometer had to sit atop of it. With the other two beakers, the thermometers were touching the bottom of the beakers. Also, with the beaker filled with water, the thermometer may have been measuring the temperature of the water (since the bulb was submerged) instead of the temperature of the air surrounding it. This may explain why the atmosphere with water showed the slowest reaction to heat. To improve this experiment next time, the experiment should include stands to hold the thermometers the same distance away from the light and off of the materials. This would help improve temperature recordings to make a more accurate experiment.